Sex, Gender, & Intersectional Analysis
Case Studies
- Science
- Health & Medicine
- Chronic Pain
- Colorectal Cancer
- Covid-19
- De-Gendering the Knee
- Dietary Assessment Method
- Gendered-Related Variables
- Heart Disease in Diverse Populations
- Medical Technology
- Nanomedicine
- Nanotechnology-Based Screening for HPV
- Nutrigenomics
- Osteoporosis Research in Men
- Prescription Drugs
- Systems Biology
- Engineering
- Assistive Technologies for the Elderly
- Domestic Robots
- Extended Virtual Reality
- Facial Recognition
- Gendering Social Robots
- Haptic Technology
- HIV Microbicides
- Inclusive Crash Test Dummies
- Human Thorax Model
- Machine Learning
- Machine Translation
- Making Machines Talk
- Video Games
- Virtual Assistants and Chatbots
- Environment
Osteoporosis Research in Men: Rethinking Standards and Reference Models
The Challenge
Men account for nearly a third of osteoporosis-related hip fractures in Europe and the U.S. (Dhanwal et al., 2010). Nonetheless, osteoporosis is considered primarily a disease of postmenopausal women, and men are rarely evaluated or treated for it (Szulc et al., 2012).
Method: Rethinking Standards and Reference Models
Research in many fields—for example, heart disease—has relied on reference models that treat men as the norm. Women are often studied as deviations from that norm. In the case of osteoporosis, however, diagnostic models have been developed for women using bone mineral density (BMD) norms of healthy young white women, and criteria to identify risk in men are not well established. Researchers are improving these reference models and opening new areas of research by considering disease progression in both women and men, and by evaluating risk using sex-specific reference models.
Gendered Innovations:
- Establishing Male Reference Populations By 1997, evaluation of men's bone quality was based on BMD norms of healthy young men rather than healthy young women (Looker, 1997). More work needs to be done to redefine diagnostic cutoffs for both women and men (Binkley et al., 2010).
- Creating New Diagnostics Based on Secondary Contributors to Osteoporosis and Metabolic Bone Disorders (SECOBs) Researchers have identified medical conditions (such as hypogonadism and hypercalciuria) and treatments (such as chemotherapeutics and anticonvulsants) that correlate with osteoporotic fracture, especially in men. New diagnostics take SECOBs into account—along with variables such as BMD, sex, and lifestyle. Accounting for these factors improves diagnosis in both women and men.
Method: Rethinking Concepts
Gendered Innovation 1: Male Reference Populations
Method: Rethinking Standards and Reference Models
Is the T≤ -2.5 Cutoff Appropriate for Male Patients?
The Male Reference Model and Osteoporosis Intervention
Method: Intersectional Approaches—Environment and Geographical Location (Differences among Men)
Ancestry, Ethnicity, and Geography
Method: Analyzing Gender - Physical Activity (Differences among Women)
Gendered Innovation 2: New Diagnostics Based on Secondary Contributors to Osteoporosis and Metabolic Bone Disorders (SECOBs)
Conclusions
Next Steps
The Challenge
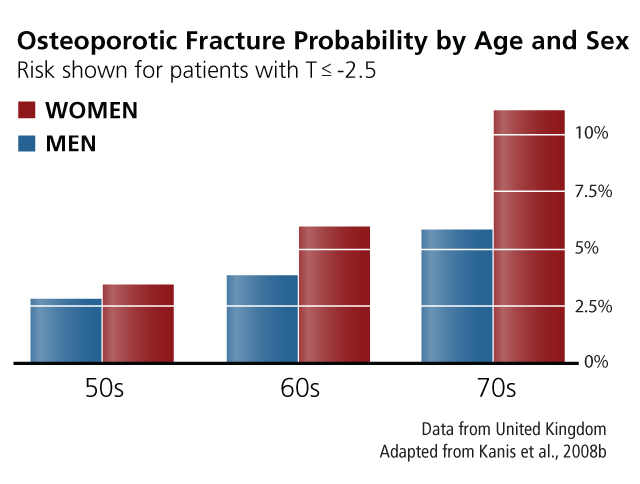
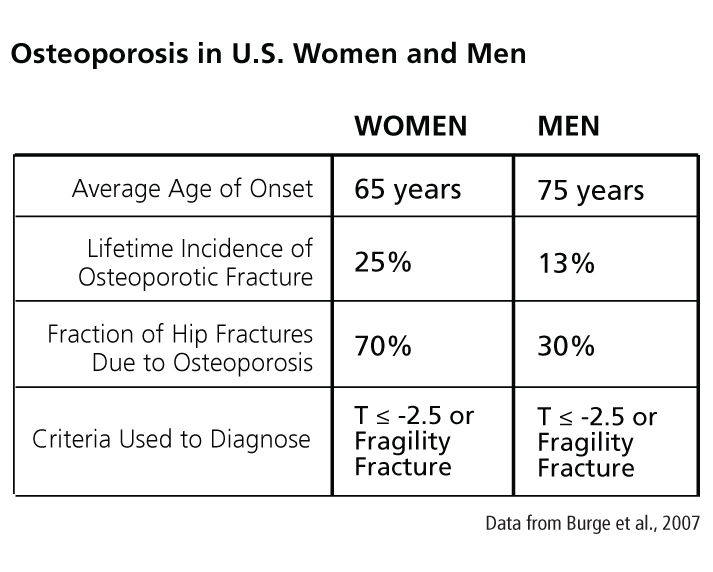 Osteoporosis has been considered primarily a disease of postmenopausal women, an assumption that has shaped its screening, practice, diagnosis, and treatment (Klinge, 2010). This perception may exist because osteoporosis manifests about 10 years earlier in women than in men (see chart at right) and because women of all ages have higher risks of fracture than age-matched men (see chart below).
Osteoporosis has been considered primarily a disease of postmenopausal women, an assumption that has shaped its screening, practice, diagnosis, and treatment (Klinge, 2010). This perception may exist because osteoporosis manifests about 10 years earlier in women than in men (see chart at right) and because women of all ages have higher risks of fracture than age-matched men (see chart below).
Although women have a higher fracture risk at a given age, medical outcomes of fractures are worse in men. A low-trauma ("fragility") fracture is associated with approximately twice the risk of a future fracture for a woman, but more than three times the risk for a man (Adler, 2018). In addition, a fragility fracture is associated with a twofold increase in mortality for a woman but a threefold increase for a man (Bliuc et al., 2009). These findings have led researchers to redefine osteoporosis as a disease affecting both women and men (see Method).
Method 1: Rethinking Concepts
Osteoporosis has traditionally been defined as a disease of white, postmenopausal women. Men, however, account for nearly a third of osteoporosis-related hip fractures in Europe and the U.S., and it is becoming clear that they have been underdiagnosed because of the limited scope of diagnostic definitions (Amin, 2010). In 2002, the U.S. Centers for Disease Control and Prevention (CDC) noted that, because of the lack of data, "there is no consensus at this time concerning the definition of low bone density in groups other than white women; however, it is clear that osteoporosis is not solely a disease of white women" (CDC, 2002). Redefining osteoporosis to include men as well as at-risk minority groups has led to new research and clinical practices that consider osteoporosis in broader populations.
View General Method
Gendered Innovation 1: Male Reference Populations
Low bone mineral density (BMD) has long been recognized as an important predictor of fracture risk. Diagnostic criteria for osteoporosis were initially based on how many standard deviations a patient's BMD drops from the mean BMD of a female reference group, specifically young (aged 20-29 years) white women (CDC, 2002). The T-score is the standard deviation from the mean for this reference group. Negative T-scores indicate bone loss. Based on studies of women, a T-score of -2.5 (i.e., 2.5 S.D. below the mean for the reference group) has been defined as the diagnostic cutoff for osteoporosis (World Health Organization, 2003).Between 1988 and 1994, the U.S. CDC collected BMD data from more than 14,000 U.S. women and men (National Center for Health Statistics, 1994). In 1997, a reference population of young men was used to calculate T-scores for male patients on the basis of healthy male (rather than healthy female) BMD values (Looker, 1997).
Calculating men's T-scores based on a male reference population greatly alters diagnosis rates. The prevalence of osteoporosis in men was estimated as 1%-4% using a female reference population; it has been estimated to be 3%-6% using a male reference population (Looker, 1997; Cummings, 2002). This practice marks an important gendered innovation (see Method).
Method : Rethinking Standards and Reference Models
When establishing reference models, researchers must consider:
In spite of these advances, problems persist. Diagnostic models based on BMD alone do not reliably predict who will suffer an osteoporotic fracture (Kanis et al., 2008a). View General Method
- The Reference Population: To what group of young, healthy people should a given patient be compared? Is it important that the patient be matched to a reference population of the same sex? The same ethnicity? Lifestyle? Geographic location?
- The Diagnostic Cutoff: How many standard deviations below the mean of the reference population best diagnose osteoporosis?
Is the T ≤ -2.5 Cutoff Appropriate for Male Patients?
The T ≤ -2.5 cutoff (using a male reference population) is a common diagnostic for osteoporosis in men, although this is a matter of debate (Szulc et al., 2012). In a recent study of more than 7,000 men and women age 55 and older, 56% of non-vertebral fractures in women and 79% of non-vertebral fractures in men occurred in participants who were not diagnosed with osteoporosis according to the T ≤ -2.5 cutoff (Schuit et al., 2004). Moreover, there are concerns about the usefulness of T-scores in predicting fracture risk, especially in premenopausal women and men under age 50 (Leslie et al., 2006; Cummings, 2006).
Multiple international models for diagnosing osteoporosis have been established. The Canadian Medical Association, the United Kingdom's National Osteoporosis Guideline Group (NOGG) and Royal College of Physicians, and the German Dachverband Osteologie e.V. (DVO) each endorse different protocols for osteoporosis diagnosis (Papaioannou et al., 2010; Compston et al., 2008; Baum et al., 2008).
The Male Reference Model and Osteoporosis Intervention
Developing a male reference population represented a gendered innovation that led, in turn, to further clinical research—see Designing Health & Biomedical Research. These include:
- Considering Bone Health as an Integral Part of Men's Health Research is underway on possible lifestyle strategies for preventing osteoporosis in men, such as a healthy diet, physical activity, and not smoking tobacco (Pinheiro et al., 2009)—see Method.
- Testing Pharmaceutical Treatments in Men Bisphosphonates, a class of anti-osteoporotic drugs, were evaluated two decades ago in postmenopausal women, but only recently in men (Francis, 2007). Including men in osteoporosis drug research may be important. Recent studies in postmenopausal women have called into question the benefits of long-term (beyond 3-5 years) bisphosphonate therapy, and the FDA now recommends that "all patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis" (Whitaker et al., 2012). More research is needed to understand the risks and benefits of specific dosing regimens in men and in pre-menopausal women (Giusti et al., 2010).
Method: Intersectional Approaches—Environment and Geographical Location (Differences among Men)
Significant differences exist between individuals of the same sex and, ostensibly, the same race. For example, widely used BMD reference values for white U.S. men have proven inappropriate for white Danish men (Høiberg et al., 2007).
An important step toward more comprehensive diagnostic criteria is the U.S. National Institutes of Health (NIH) Study of Osteoporotic Fractures in Men ("Mr. OS"), which began enrolling a cohort of 6,000 U.S. men 65 years and older in 2000 and was extended to include large cohorts of men in China and in Sweden. In addition to examining the relation between BMD and fracture risk in men, Mr. OS examines factors—exercise level, diet, body composition, tobacco use, and alcohol—that often correlate with sex, race, and ethnicity (Bennett, 2004; Cauley et al., 2005).
View General Method
Ancestry, Ethnicity, and Geography
For patients of a given sex, factors such as ancestry and ethnicity should also be considered in establishing reference populations. In the U. S., significant differences are seen in fracture risk between women of different self-reported races. Although African American women have lower fracture rates than white women (48% lower risk), black women have higher mortality after hip fracture than do white women. Reasons may include socioeconomic disparities, unequal access to treatment, and prevalence of other diseases (Thomas, 2007)—see Method.
Method: Analyzing Gender—Physical Activity (Differences among Women)
Biologist Anne Fausto-Sterling has described how environment and experience can "shape the very bones that support us." Osteoporosis is a complex disease that emerges over the lifecycle as a response to "specific lived lives" (Fausto-Sterling, 2008). Gender roles interact with sex in determining bone strength: In Europe and the U.S., adolescent girls may exercise less than boys. Along with biological factors, these gendered behaviors result in girls laying down less bone than boys in their teens. In addition, occupational divisions of labor mean that men are more likely than women to do heavy physical work, such as construction (Fausto-Sterling, 2005). And older women are generally less physically active than their male counterparts; inactivity may contribute to bone loss and increase fracture risk. View General Method
Gendered Innovation 2: New Diagnostics Based on Secondary Contributors to Osteoporosis and Metabolic Bone Disorders (SECOBs)
Researchers studying differences both between and within groups of women and men have identified secondary contributors to osteoporosis and metabolic bone disorders (SECOBs), medical conditions, and treatments that increase the risk of osteoporotic fracture. Understanding SECOBs is especially important in estimating men's fracture risk; men with fragility fractures are more likely than women to have previously diagnosed SECOBs, and when patients are screened after a fracture, new SECOBs are more often found in men (50%) than in women (32%) (Ryan et al., 2011; Tannenbaum et al., 2002). New diagnostics include the following:
| Diagnostic Tool | Developer | Reference Population | Covariates Used to Calculate Fracture Risk | |||
FRAX |
WHO Collaborating Centre for Metabolic Bone Diseases, University of Sheffield, UK (WHO, 2003). |
46,340 patients (32% men) from 19 EU countries, Australia, Canada, Japan, and the US (Kanis et al., 2007). |
Demographics:
Diagnostic Measurements:
Patient Characteristics: | |||
| Garvan Fracture Risk Calculator (GFRC) | Osteoporosis and Bone Biology Program, Garvan Institute of Medical Research, Sydney (Sandhu et al., 2010). | 2,216 patients (39% men, 98.6% Caucasian) from Dubbo, Australia (Nguyen et al., 2008). | Demographics: -Age (patients 60 and older only) -Sex
Diagnostic Measurements: | |||
| QFracture | QResearch Scientific Board (Hippisley-Cox et al., 2009). | 2,357,895 patients (49% men) from England and Wales (Hippisley-Cox et al., 2009). | Demographics: -Age (patients 35-84 only) -Sex
Patient Characteristics (used for evaluating
women and men):
Patient Characteristics (used for evaluating
women only): |
In addition to the above, other systems of fracture risk estimation exist; Gimeno et al. describe nine different systems as "most commonly used" (2010). Clinicians continue to debate the relative advantages and disadvantages of the FRAX, GFRC, and QFracture systems, all of which can be used to estimate a patient's risk of osteoporotic fracture over the next ten years (Bolland et al., 2011; Bennett et al., 2010).
Conclusions
Osteoporosis has been reconceptualized as affecting both women and men. This gendered innovation led to the development of male reference populations, allowing for better evaluation of fracture risk in men. BMD alone, however, is not a good predictor of fracture in women or men. New diagnostics, such as FRAX, GFRC, and QFracture, may predict a patient's fracture risk more accurately than BMD alone by analyzing factors intersecting with sex and gender.
Next Steps
- Use gender analysis to optimize osteoporosis prevention. Many risk factors and protective factors are gendered. A prevention campaign might focus on increasing physical activity and on smoking cessation, recognizing that women are more likely to be physically inactive and men are more likely to smoke tobacco.
- Develop reference models focused on how fracture risk is influenced by biological sex and gendered behaviors. The U.S. Preventive Services Task Force has concluded that "evidence is lacking, of poor quality, or conflicting" regarding osteoporosis screening for men (USPSTF, 2011). Current European guidelines for the diagnosis and management of osteoporosis recommend evaluating men's fracture risk according to diagnostic thresholds developed to predict fracture risk in postmenopausal women (Kanis et al., 2008b).
- Work to educate the public about the true incidence of the disease and to promote bone-healthy lifestyles in women and men (NIH, 2010). The NIH has determined through surveys that a majority of American men view osteoporosis as a "woman's disease". Correcting this inaccurate view is important. The gendered beliefs of physicians may also contribute to the perception that osteoporosis is a woman's disease, resulting in osteoporosis in men being "substantially underdiagnosed, undertreated, and underreported" (Qaseem et al., 2008; Geusens et al., 2007).
Works Cited
Adler, R. A. (2018). Update on Osteoporosis in Men. Best Practice & Research Clinical Endocrinology & Metabolism, 32(5), 759-772.
Ahmed, A.I.H., Blake, G.M., Rymer, J.M., & Fogelman, I. (1997). Screening for Osteopenia and Osteoporosis: Do the Accepted Normal Ranges Lead to Overdiagnosis? Osteoporosis International, 7, 432-438.
Aloia, J.F. (2003). African Americans, 25-hydroxyvitamin D, and osteoporosis: a paradox. The American Journal of Clinical Nutrition, 88 (supplement), 545S-50S.
Amin, S. (2010). Epidemiology of Fractures. In Orwoll, E., Bilezikian, J., & Vanderschueren, D. (Eds.), Osteoporosis in Men: The Effects of Gender on Skeletal Health, Second Edition, pp. 351-361. London: Elsevier.
Amin, S., Zhang, Y., Felson, D.T., Sawin, C.T., Hannan, M.T., Wilson, P.W., & Kiel, D.P. (2006). Estradiol, Testosterone, and the Risk for Hip Fractures in Elderly Men from the Framingham Study. American Journal of Medicine, 119, 426-433.
Baron, J.A., Barrett, J., Malenka, D., Fisher, E., Kniffin, W., Bubolz, T., & Tosteson, R. (1994). Racial Differences in Fracture Risk. Epidemiology, 1 (5), 42-47.
Bennett, B. (2004). Osteoporosis in Men. The NIH Word on Health: Consumer Health Information Based on Research from the National Institutes of Health, April.
Bennett, A., Adlan, A., & Johansen, A. (2010). FRAX and QFracture—Which is the Better Screening Tool for Hip Fracture Risk? Osteoporosis International: Abstracts of the Osteoporosis Conference 2010, 21 (3), S447.
Binkley, N., & Alder, R. (2010). Dual-Energy X-Ray Absorptiometry (DXA) in Men. In Orwoll, E., Bilezikian, J., & Vanderschueren, D. (Eds.), Osteoporosis in Men: The Effects of Gender on Skeletal Health, Second Edition, pp. 525-541. London: Elsevier.
Binkley, N.C., Schmeer, P., Wasnich, R.D., & Lenchik, L. (2002). What Are the Criteria by Which a Densitometric Diagnosis of Osteoporosis Can Be Made in Males and Non-Caucasians? Journal of Clinical Densitometry, 3 (5), s19-s27.
Burge, R., Dawson-Hughes, B., Solomon, D., Wong, J., King, A., & Tosteson, A., (2007). Incidence and Economic Burden of Osteoporosis-Related Fractures in the United States, 2005-2025. Journal of Bone and Mineral Research, 22 (3), 465-475.
Campion, J.M., & Maricic, M.J. (2003). Osteoporosis in Men. American Family Physician, 7 (67), 1521-1526.
Castro, J.P., Joseph, L.A., Shin, J.J., Arora, S.K., Nicasio, J., Shatzkes, J., Raklyar, I., Erlikh, E., Pantone, V., Bahtiyar, G., Chandler, L., Pabon, L., Choudhry, S., Ghadiri, N., Pramodini, G., Muniyappa, R., von-Gicyzki, H., & McFarlane, S.I. (2005). Differential Effect of Obesity on Bone Mineral Density in White, Hispanic and African American Women: A Cross Sectional Study. Nutrition and Metabolism, 9 (2), 1-7.
Cauley, J., Fullman, R.L., Stone, K.L., Zmuda, J.M., Bauer, D.C., Barrett-Connor, E., Ensrud, K., Lau, E., & Orwoll, E.S. (2005). Factors Associated with the Lumbar Spine and Proximal Femur Bone Mineral Density in Older Men. For the Mr. OS Research Group. Osteoporosis International, 16, 1525-1537.
Cauley, J.A., Zmuda, J.M., Wisniewski, S.R., Krishnaswami, S., Palermo, L., Stone, K.L., Black, D.M., & Nevitt, M.C. (2004). Bone Mineral Density and Prevalent Vertebral Fractures in Men and Women. Osteoporosis International, 15, 32-37.
Centers for Disease Control and Prevention. (2002). National Health and Nutrition Examination Survey (NHANES): Osteoporosis. Washington, D.C.: United States Department of Health and Human Services National Center for Health Statistics.
Cummings, S.R., Bates, D., & Black, D.M. (2002). Clinical Use of Bone Densitometry: Scientific Review. Journal of the American Medical Association, 16 (288), 1889-1897.
Cummings, S.R., Cawthon, P.M., Ensrud, K.E., Cauley, J.A., Fink, H.A., & Orwoll, E.S. for the Osteoporotic Fractures in Men (MrOS) and Study of Osteoporotic Fractures (SOF) Research Groups. (2006). BMD and Risk of Hip and Nonvertebral Fractures in Older Men: A Prospective Study and Comparison With Older Women. Journal of Bone and Mineral Research, 10 (21), 1550-1556.
Cummings, S.R., Cauley, J.A., Palermo, L., Ross, P.D., Wasnich, R.D., Black, D., & Falkner, K.G. for the Study of Osteoporotic Fractures Research Group. (1994). Racial Differences in Hip Axis Lengths Might Explain Racial Differences in Rates of Hip Fracture. Osteoporosis International, 4, 226-229.
Durie, B., Katz, M., Crowley, J. (2005). Osteonecrosis of the Jaw and Bisphosphonates. New England Journal Of Medicine, 353, 99-102.
Duster, T. (2009). Debating Reality and Relevance. Science, 324 (5931), 1144-1145.
Ebeling, P.R. (2008). Osteoporosis in Men. The New England Journal of Medicine, 358, 1474-1482.
Farmer, M.E., White, L.R., Brody, J.A., & Bailey, K.R. (1984). Race and Sex Differences in Hip Fracture Incidence. American Journal of Public Health, 12 (74), 1374-1380.
Faulkner, K. & Orwoll, E. (2002). Implications in the Use of T-Scores for the Diagnosis of Osteoporosis in Men. Journal of Clinical Densitometry, 1 (5), 87-93.
Fausto-Sterling, A. (2008). The Bare Bones of Race. Social Studies of Science, 38, 657-694.
Fausto-Sterling, A. (2005). The Bare Bones of Sex: Part 1 – Sex and Gender. Signs: Journal of Women in Culture and Society, 2 (30), 1491-1527.
Finkelstein, J.S. Brockwell, S.E., Mehta, V., Greendale, G.A., Sowers, M.F.R., Ettinger, B., Lo, J.C., Johnston, J.M., Cauley, J.A., Danielson, M.E., & Neer, R.M. (2008). Bone Mineral Density Changes During the Menopause Transition in a Multiethnic Cohort of Women. The Journal of Clinical Endocrinology and Metabolism, 93, 861-868.
Fleming, R. (1999). New NIAMS Grant Targets Osteoporosis in Men. National Institutes of Health.
Francis, M.D., & Valent, D.J. (2007). Historical Perspectives on the Clinical Development of Bisphosphonates in the Treatment of Bone Diseases. Journal of Musculoskeletal and Neuronal Interactions, 1 (7), 2-8.
Geusens, P., & Dinant, G. (2007). Integrating a Gender Dimension into Osteoporosis and Fracture Risk Research. Gender Medicine, 4 (S2), S147-S161.
Giusti, A., & Papapoulos, S. (2010). Treatment of Male Osteoporosis with Bisphosphonates. In Orwoll, E., Bilezikian, J., & Vanderschueren, D. (Eds.), Osteoporosis in Men: The Effects of Gender on Skeletal Health, Second Edition, pp. 667-681. London: Elsevier.
Griffin, M.R., Ray, W.A., Fought, R.L., & Melton III, L.J. (1992). Black-White Differences in Fracture Rates. American Journal of Epidemiology, 11 (36), 1378-1385.
Gürlek, A., Bayraktar, M., & Ariyürek, M. (2000). Inappropriate Reference Range for Peak Bone Mineral Density in Dual-Energy X-Ray Absorptiometry: Implications for the Interpretation of T-scores. Osteoporosis International, 11, 809-813.
Høiberg, M., Nielsen, T.L., Wraae, K., Abrahamsen, B., Hagen, C., Andersen, M., & Brixen, K. (2007). Population-Based Reference Values for Bone Mineral Density in Young Men. Osteoporosis International, 18, 1507-1514.
Jackson, K.A., & Savaiano, D.A., (2001). Lactose Maldigestion, Calcium Intake and Osteoporosis in African-, Asian-, and Hispanic-Americans. Journal of the American College of Nutrition, 2 (20), 198S-207S.
Jacobsen, S.J., Goldberg, J., Miles, T.P., Brody, J.A., Stiers, W., & Rimm, A.A. (1990). Regional Variation in the Incidence of Hip Fracture. Journal of the American Medical Association, 4 (264), 500-502.
Kanis, J., Burlet, N., Cooper, C., Delmas, P., Reginster, J., Borgstrom, F., & Rizzoli, R. (2008a) European Guidance for the Diagnosis and Management of Osteoporosis in Postmenopausal Women. Osteoporosis International, 19 (4), 399-428.
Kanis, J., Johnell, O., Oden, A., & McCloskey, E. (2008b). FRAX and the Assessment of Fracture
Probability in Men and Women from the UK. Osteoporosis International, 19, 385-397.
Kanis, J., Oden, A., Johnell, O., Johansson, H., De Laet, C., Brown, J., Burckhardt, P., Cooper, C., Christiansen, C., Cummings, S., Eisman, J., Fujiwara, S., Glüer, C., Goltzman, D., Hans, D., Krieg, M., La Croix, A., McCloskey, E., Mellstrom, D., Melton, L., Pols,. H., Reeve, J., Sanders, K., Schott, A., Silman, A., Torgerson, D., van Staa, T., Watts, N., & Yoshimura, N. (2007). The Use of Clinical Risk Factors Enhances the Performance of BMD in the Prediction of Hip and Osteoporotic Fractures in Men and Women. Osteoporosis International, 18 (8), 1033-1046.
Karagas, M.R., Lu-Yao, G.L., Barrett, J.A., Beach, M.L., & Baron, J.A. (1996). Heterogeneity of Hip Fracture: Age, Race, Sex, and Geographic Patterns of Femoral Neck and Trochanteric Fractures among the US Elderly. American Journal of Epidemiology, 7 (143), 677-682.
Klinge, I. (2010). Innovative Changes in Biomedicine: Integration of Sex and Gender Aspects in Research and Clinical Practice. GenderChange in Academia, 10, 231-242.
LeBlanc, E.S., Nielson, C.M., Marshall, L.M., Lapidus, J.A., Barrett-Connor, E., Ensrud, K.E., Hoffman, A.R., Laughlin, G., Ohlsson, C., & Orwoll, E.S. (2009). The Effects of Serum Testosterone, Estradiol, and Sex Hormone Binding Globulin Levels on Fracture Risk in Older Men. Journal of Clinical Endocrinology & Metabolism, 94, 3337-3346.
Lei, S.F., Deng, F.Y., Liu, X.H., Huang, Q.R., Qin, Y., Zhou, Q., Jiang, D.K., Li, Y.M., Mo, X.Y., Liu, M.Y., Chen, X.D., Wu, X.S., Shen, H., Dvornyk, V., Zhao, L., Recker, R.R., & Deng, H.W. (2003). Polymorphisms of Four Bone Mineral Density Candidate Genes in Chinese Populations and Comparison with Other Populations of Different Ethnicity. Journal of Bone and Mineral Metabolism, 21, 34-42.
Looker, A.C., Orwoll, E.S., Johnston, C.C. Jr., Lindsay, R.L., Wahner, H.W., Dunn, W.L., Calvo, M.S., Harris, T.B., & Heyse, S.P. (1997). Prevalence of Low Femoral Bone Density in Older U.S. Adults from NHANES III. Journal of Bone and Mineral Research, 11 (12), 1761-8.
Meier, C., Nguyen, T.V., Handelsman, D.J., Schindler, C., Kushnir, M.M., Rockwood, A.L., Meikle, A.W., Center, J.R., Eisman, J.A., & Seibel, M.J. (2008). Endogenous Sex Hormones and Incident Fracture Risk in Older Men. Archives of Internal Medicine, 168, 47-54.
Mellström, D., Vandenput, L., Mallmin, H., Holmberg, A.H., Lorentzon, M., Odén, A., Johansson, H., Orwoll, E.S., Labrie, F., Karlsson, M.K., Ljunggren, O., & Ohlsson, C. (2008). Older Men with Low Serum Estradiol and High Serum SHBG Have an Increased Risk of Fractures. Journal of Bone Mineral Research, 23, 1552-1560.
Melton, L.J. 3rd, Marquez, M.A., Achenbach, S.J., Tefferi, A., O’Connor, M.K., O’Fallon, W.M., & Riggs, B.L. (2002). Variations in Bone Density Among Persons of African Heritage. Osteoporosis International, 7 (13), 551-559.
Melton, L.J. 3rd. (2001). The Prevalence of Osteoporosis: Gender and Racial Comparison. Calcified Tissue International, 69 (4), 179-81.
Melton, L.J. 3rd, Khosla, S., Achenbach, S.J., O’Connor, M.K., O’Fallon, W.M., & Riggs, B.L. (2000). Effects of Body Size and Skeletal Site on the Estimated Prevalence of Osteoporosis in Women and Men. Osteoporosis International, 11 (11), 977-983.
Miller, P.D. (2006). Guidelines for the Diagnosis of Osteoporosis: T-scores vs Fractures. Reviews in Endocrine & Metabolic Disorders, 7, 75-89.
Orwoll, E.S. (2009). Abstract—Grant Number: 5U01AG027810-03, Project Title: Osteporotic [sic] Fractures in Men (MrOS)-Admin Center. Computer Retrieval of Information on Scientific Projects (CRISP). National Institutes of Health. Retrieved 28 February 2009 from
Orwoll, E., Blank, J.B., Barrett-Connor, E., Cauley, J., Cummings, S., Ensrud, K., Lewis, C., Cawthon, P.M., Marcus, R., Marshall, L.M., McGowan, J., Phipps, K., Sherman, S., Stefanick, M.L., & Stone, K. (2005). Design and Baseline Characteristics of the Osteoporotic Fractures in Men (MrOS) Study—A Large Observational Study of the Determinants of Fracture in Older Men. Contemporary Clinical Trials, 26, 569-585.
Qaseem, A., Snow, V., Shekelle, P., Hopkins, R. Jr., Forciea, M.A., & Owens, D.K. (2008). Screening for Osteoporosis in Men: A Clinical Practice Guideline from the American College of Physicians. Annals of Internal Medicine, 9 (148), 680-684.
Sweet, M.G., Sweet, J.M., Jeremiah, M.P., & Galazka, S.S. (2009). Diagnosis and Treatment of Osteoporosis. American Family Physician, 3 (79), 193-200.
Szulc, P., Kaufman, J., & Orwoll, E. (2012). Osteoporosis in Men. Journal of Osteoporosis, 1-5.
Whitaker, M., Guo, J., Kehoe, T., & Benson, G. (2012). Bisphosphonates for Osteoporosis--Where Do We Go from Here? New England Journal of Medicine, online in advance of print.
Wong, S.Y.S., Lau, E.M.C., Lynn, H., Leung, P.C., Woo, J., Cummings, S.R., & Orwoll, E. (2004). Depression and Bone Mineral Density: Is There A Relationship in Elderly Asian Men? Results From Mr. OS (Hong Kong). Osteoporosis International, 6 (16), 610-615.
Wiemann, L.M., Vallarta-Ast, N., Krueger, D., & Binkley, N. (2007). Effect of Female Database Use for T-score Derivation in Men. Journal of Clinical Densitometry, 2 (9), 238.
The World Health Organization. (2003). Prevention and Management of Osteoporosis: Report of a WHO Scientific Group. Geneva, Switzerland.
Wu, X.P., et al. (2003). A Comparison Study of the Reference Curves of Bone Mineral Density at Different Skeletal Sites in Native Chinese, Japanese, and American Caucasian Women. Calcified Tissue International, 73, 122-132.
Osteoporosis has long been defined as a disease primarily of post-menopausal women—an assumption that has shaped its screening, diagnosis, and treatment. Why is this a problem? Men account for a third of osteoporosis-related hip fractures after age 75—and when they break their hips, they die more often than women. We don't know why.
Despite the relatively high number of men who suffer from osteoporosis, the basic diagnostics for the disease were developed using young, white women (aged 20-29 years).
Gendered Innovation:
The breakthrough came in 1997 when a reference population of young men was established to diagnose osteoporosis in men. Although we now have reference populations for men, men are still diagnosed using the female diagnostic cut-off rate—this has not yet been revised for men.
Work continues to diagnose osteoporosis in different populations of women and men. Osteoporosis is a disease with both sex and gender components: bones are formed by biology and also by culture, such as exercise rates, nutrition, and general lifestyle. These differences in lifestyle may explain differences in osteoporosis rates across ethnic groups. Current studies are analyzing cohorts of men from China and Sweden, for example, to understand these types of differences. The goal is to maintain healthy bones in diverse populations.






